Rosmarinus officinalis (leaf)
Contents |
Nomenclature
Rosmarinus officinalis L. Lamiaceae
Standardized common name (English): rosemary
Botanical Voucher Specimen
|
|
Organoleptic Characteristics
|
Macroscopic Characteristics
|
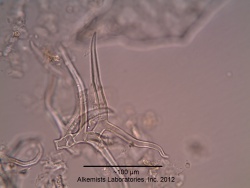
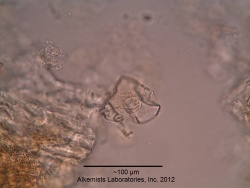
Microscopic Characteristics
 |
 |
|
|
|
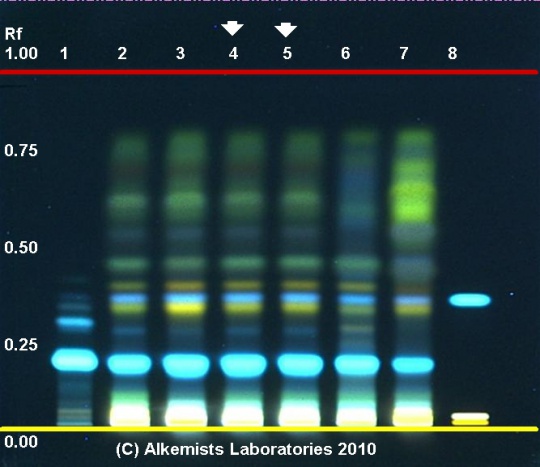
High Performance Thin Layer Chromatographic Identification
|
Rosemary (leaf) (Rosmarinus officinalis) Lane Assignments Lanes, from left to right (Track, Volume, Sample):
Reference materials used here have been authenticated by macroscopic, microscopic &/or TLC studies according to the reference source cited below held at Alkemists Laboratories, Costa Mesa, CA. Stationary Phase Silica gel 60, F254, 10 x 10 cm HPTLC plates Mobile Phase toluene: ethyl formate: HCOOH [5/4/1] Sample Preparation Method 0.3g+3mL 80% CH3OH sonicate/heat @ 50°C ~ 1/2 hr Detection Method Natural Product Reagent + PEG -> UV 365 nm Reference see British Pharmacopoeia, 2003
|
|
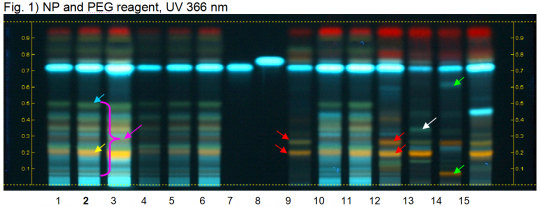
Rosemary (leaf) (Rosmarinus officinalis) Lane Assignments Lanes, from left to right (Track, Volume, Sample):
Reference Sample(s) Reference: Dissolve 5 mg of rosmarinic acid in 10 mL of methanol. Dissolve 6 mg of caffeic acid in 10 mL of methanol. Stationary Phase Stationary phase, i.e. Silica gel 60, F254 Mobile Phase Ethyl acetate, formic acid, water 15:1:1 (v/v/v) Sample Preparation Method Sample: Mix 1 g of powdered sample with 10 mL of methanol and sonicate for 10 minutes, then centrifuge or filter the solutions and use the supernatants / filtrates as test solutions. Derivatization reagent: 1.) NP reagent Preparation: 1 g of natural products reagent in 200 mL of ethyl acetate 2.) PEG reagent Preparation: 10 g of polyethylene glycol 400 in 200 mL of dichloromethane Use: Heat plate 3 min at 100°C, then dip (time 0, speed 5) in NP reagent, dry and dip (time 0, speed 5) in PEG reagent Detection Method Saturated chamber; developing distance 70 mm from lower edge; relative humidity 33% Other Notes Images presented in this entry are examples and are not intended to be used as basis for setting specifications for quality control purposes. System suitability test: Rosmarinic acid: light blue fluorescent zone at Rf ~ 0.73. Caffeic acid: light blue fluorescent zone at Rf ~ 0.76. Identification: Compare result with reference images. The fingerprint of the test solution is similar to that of the corresponding botanical reference sample. Additional weak zones may be present. The chromatogram of the test solution shows a red zone right below the solvent front. At the position of reference substance rosmarinic acid an intense light blue zone is seen. A green zone is detected at Rf ~ 0.50 (blue arrow). Below this zone there is a pattern of characteristically colored zones of low intensity (pink arrow). An intense orange zone is detected at Rf ~ 0.21 (yellow arrow). Test for other species: There are no two orange zones at Rf ~ 0.27 and 0.20 (red arrows, Oregano leaf or Thyme leaf). No light blue zone is seen at Rf ~ 0.32 (white arrow, Holy basil leaf). No orange zone is seen at Rf ~ 0.27 and no zone is seen at Rf ~ 0.63 (green arrows, Sweet basil leaf).
|
Supplementary Information
Sources
- ↑ MOBOT, Tropicos.org http://www.tropicos.org/Image/100002347
- ↑ United States Dispensatory (1918)
- ↑ United States Dispensatory (1918)
- ↑ PlantaPhile http://plantaphile.com/
- ↑ PlantaPhile http://plantaphile.com/
- ↑ Elan M. Sudberg, Alkemist Laboratories http://www.alkemist.com
- ↑ Elan M. Sudberg, Alkemist Laboratories http://www.alkemist.com
- ↑ Elan M. Sudberg, Alkemist Laboratories http://www.alkemist.com
- ↑ HPTLC Association http://www.hptlc-association.org/