Achillea millefolium (flower)
Contents |
Nomenclature
Achillea millefolium L. Asteraceae
Standardized common name (English): yarrow
Botanical Voucher Specimen
 |
 |
|
|
|
|
|
Organoleptic Characteristics
Macroscopic Characteristics
|
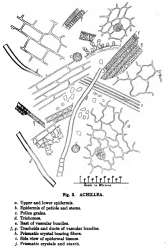
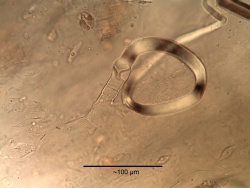
Microscopic Characteristics
|
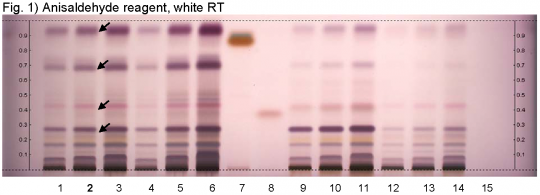
High Performance Thin Layer Chromatographic Identification
|
Yarrow (flower) (Achillea millefolium) Lane Assignments Lanes, from left to right (Track, Volume, Sample):
Reference Sample(s) Reference: Dissolve 25 mg of cineole in 20 mL of tolulene. Dissolve 10 mg of guaiazulene in 20 mL of tolulene. Stationary Phase Stationary phase, i.e. Silica gel 60, F254 Mobile Phase Tolulene, ethyl acetate 95:5 (v/v) Sample Preparation Method Sample: Mix 1.0 g of powdered sample with 10 mL of methanol and sonicate for 10 minutes, then centrifuge or filter the solutions and use the supernatants / filtrates as test solutions. Derivatization reagent: Anisaldehyde reagent; Preparation: 170 mL of ice cooled methanol are mixed with 20 mL of acetic acid, 10 mL of sulfuric acid, and 1 mL of anisaldehyde. Use: Dip (time 0, speed 5), heat at 100°C for 4 min. Detection Method Saturated chamber; developing distance 70 mm from lower edge; relative humidity 33% Other Notes Images presented in this entry are examples and are not intended to be used as a bases for setting specifications for quality control purposes. System suitability test: Cineole: violet zone at Rf ~ 0.38. Guaiazulene: brown zone at Rf ~ 0.88. Application: 2 µL of references, 16 µL of test solutions Identification: Compare result with reference images. The fingerprint of the test solution is similar to that of the corresponding botanical reference sample. Additional weak zones may be present. The chromatogram of the test solution shows a violet zone at Rf ~ 0.94 right above the zone due to guaiazulene reference substance. Below this zone there is a violet zone at Rf ~ 0.70. There is a reddish-violet zone at Rf ~ 0.43 right above the zone due to cineole reference substance. Below this zone there is a violet zone at Rf ~ 0.29 (black arrows).
|
Supplementary Information
CE/HPLC flavonoid determination, Kocevar, et al., 2008
Comparison of capillary electrophoresis and high performance liquid chromatography for determination of flavonoids in Achillea millefolium,
Abstract. Flavonoids represent an important bioactive component in Achillea millefolium. The comparison of the most commonly used analytical methods for the identification and quantification of flavonoids, capillary electrophoresis (CE) and high performance liquid chromatography (HPLC), is presented. The methods were optimized and validated. Using a 20 mM borate buffer with 30% (v/v) of methanol (pH 9.3) in the CE analysis and a gradient elution with water–acetonitrile mobile phase in the HPLC analysis, sufficient separation of the analytes was achieved. A relatively high injection volume in the CE analysis (30 mbar × 30 s) enabled low limit of detection (LOD) (0.3–0.7 mg/L). Repeatability of both methods was acceptable (relative standard deviation of peak area were <6%). Additionally, the amount of flavonoids in a real sample of the dried herbal drug was determined.[9]
Sources
- ↑ Tropicos.org. Missouri Botanical Garden. 05 Aug 2013 http://www.tropicos.org/Image/88582
- ↑ Botanical Voucher Specimen Library, Alkemists Laboratories http://www.alkemist.com
- ↑ Royal Botanic Gardens, Kew. http://specimens.kew.org/herbarium/K000942133
- ↑ American Medicinal Plants of Commercial Importance (1930)
- ↑ Schneider, A. (1921) The Microanalysis of Powdered Vegetable Drugs, 2nd ed.
- ↑ Schneider, A. (1921) The Microanalysis of Powdered Vegetable Drugs, 2nd ed.
- ↑ Elan M. Sudberg, Alkemist Laboratories http://www.alkemist.com
- ↑ HPTLC Association http://www.hptlc-association.org/
- ↑ Kočevar, N., Glavač, I., Injac, R., Kreft, S., 2008. Comparison of capillary electrophoresis and high performance liquid chromatography for determination of flavonoids in Achillea millefolium Journal of Pharmaceutical and Biomedical Analysis 46(3), 609-614. http://dx.doi.org/10.1016/j.jpba.2007.11.016
- Botanical
- Asteraceae
- Media
- Voucher
- Tropicos.org. Missouri Botanical Garden. 05 Aug 2013
- Botanical Voucher Specimen Library, Alkemists Laboratories
- Royal Botanic Gardens, Kew.
- Macroscopy
- American Medicinal Plants of Commercial Importance (1930)
- Microscopy
- Schneider, A. (1921) The Microanalysis of Powdered Vegetable Drugs, 2nd ed.
- Elan M. Sudberg, Alkemist Laboratories
- HPTLC
- HPTLC Association